Abstract
Background The use of next-generation sequencing in the clinic has increasingly uncovered new variants of unknown significance (VUS), which are often ignored clinically. Ataxia telangiectasia mutated (ATM) is a tumor-suppressor gene that has a critical role in DNA double-strand break repair and has been implicated in the pathogenesis of CLL. In this study, we assessed rare germline ATM variants including VUS using a novel variant calling algorithm, evaluated their impact on the clinical characteristics of CLL and functionally assessed their effect on ATM kinase activity.
Methods We evaluated the entire ATM coding region via germline whole exome sequencing from 885 CLL patients representing three different previously reported cohorts (327 DFCI; 276 German CLL Study Group, and 282 ICGC), along with a cohort of ancestry matched controls. We utilized DeepVariant (Poplin et al Nature Biotech 2018) to detect germline variants, followed by a standardized quality control workflow (AlDubayan et al JAMA 2020). All individuals harbored ATM germline variants. Variants were annotated using dbSNP, Clinvar and Varsome, and variants with GnomAD population allele frequency (AF) < 1% were defined as rare. For clinical characterization of CLL patients with rare germline ATM missense variants, we focused on the DFCI cohort. For functional analysis, we evaluated ATM kinase activity by ATM and KAP1 phosphorylation in response to 1 Gy gamma-irradiation using flow cytometry in primary CLL samples with/without rare germline ATM missense variants. We also generated CRISPR/Cas9 engineered cell lines to evaluate the function of ATM p.L2307F which was frequent in our CLL cohort and which we previously demonstrated as enriched in CLL, yet generally annotated as a VUS.
Results We identified a total of 130 ATM germline variants among the CLL cases, including 87 missense, and 9 splice site alterations. 80 of 87 missense variants were rare and 59 were annotated as VUS. The top 5 most frequent rare germline missense variants with high odds ratio for association with CLL were ATM p.L2332P (AF cases vs controls, 0.328% vs 0.0188%, OR 17.46), p.L2307F (OR 16.88), p.F763L (OR 18.89), p.Y1442H (OR 35.39) and p.S99G (OR 16.28). In 63 DFCI patients with rare missense variants, the median age at diagnosis was 55 (31-82), 21.7% had a familial history of CLL and 29% had del(11q).
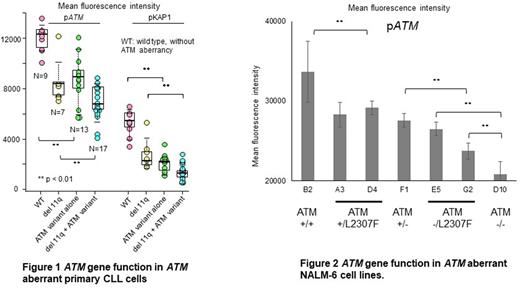
Phosphorylation of ATM and KAP1 (pATM, pKAP1) in response to gamma-irradiation was reduced in 12 patients assessed (8 rare and 4 non-rare) with germline ATM variants (ATM variant alone group) compared to those without any somatic or germline ATM aberrancy (WT group) (P<0.01). Similar results were seen when the analysis was restricted to the 8 CLL patients with rare ATM variants. Phosphorylation levels were also significantly lower in 17 patients with rare germline ATM variants with concurrent del(11q) (del 11q + ATM variant group) compared to 13 of those with del(11q) alone (del 11q group) (P<0.01) (Figure 1). Altogether, these results suggest that germline ATM variants have a functional impact on DNA damage response in primary CLL cells.
We previously identified the VUS variant ATM p.L2307F as particularly enriched among CLL patients (Tiao G et al., Leukemia. 2017). We therefore generated ATM L2307F knock-in NALM-6 cell lines via CRISPR/Cas9. We generated 7 cell lines including ATM wild type (+/+), ATM monoallelic mutated (+/L2307F), ATM monoallelic knocked out (+/-), ATM monoallelic mutated with knock out of the other allele (-/L2307F), and ATM biallelic knockout (-/-). The induction of both pATM and pKAP1 levels by gamma irradiation was reduced in proportion to ATM mutational status as shown in Figure 2.
Conclusions We demonstrate that rare ATM germline variants are common in CLL and affect the DNA damage response in primary CLL cells when controlling for somatic 11q deletion. We further characterize the p.L2307F variant using CRISPR and demonstrate that this allele represents a functional hypomorph in cell-based assays. Further studies are needed to assess the impact of each individual rare germline variant on the function of ATM, CLL pathogenesis and the inherited risk of CLL.
Disclosures
Gillani:Google: Current equity holder in publicly-traded company; Microsoft: Current equity holder in publicly-traded company; Amazon: Current equity holder in publicly-traded company; Apple: Current equity holder in publicly-traded company; Moderna: Current equity holder in publicly-traded company; Pfizer: Current equity holder in publicly-traded company; Vertex: Current equity holder in publicly-traded company; Berkshire Hathaway: Current equity holder in publicly-traded company; Target: Current equity holder in publicly-traded company; JP Morgan: Current equity holder in publicly-traded company; Goldman Sachs: Other: Minority stakes. Getz:MSMuTect: Patents & Royalties; MSMutSig: Patents & Royalties; MSIDetect: Patents & Royalties; IBM: Research Funding; SignatureAnalyzer-GPU: Patents & Royalties; Pharmacyclics: Research Funding; Scorpion Therapeutics: Consultancy, Current equity holder in publicly-traded company, Other: Founder; POLYSOLVER: Patents & Royalties. Wu:BioNTech: Current equity holder in publicly-traded company; Pharmacyclics: Research Funding. Brown:Grifols: Consultancy; TG Therapeutics: Research Funding; Bristol-Myers-Squibb/Juno/Celgene: Consultancy; Catapult: Consultancy; Hutchmed: Consultancy; Gilead: Research Funding; MEI Pharma: Consultancy, Research Funding; Sun: Research Funding; SecuraBio: Research Funding; Genentech/Roche: Consultancy; Pharmacyclics: Consultancy; Eli Lilly: Consultancy, Research Funding; Janssen: Consultancy; Abbvie: Consultancy; iOnctura: Consultancy; Beigene: Consultancy, Research Funding; Acerta/ Astra-Zeneca: Consultancy; Pfizer: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal